|
The protocol is based on the excellent instructions for using ImageJ to Below is an example of a Western Blot with six lanes showing the Tags: densitometry, Gel, gel quantification, ImageJ, Western blot the Gel Analysis method outlined in the ImageJ documentation: Gel Analysis This is what you get when you treat each row in the dot blot as a horizontal "lane" and use the gel analysis procedure in the ImageJ manual. Use Reset Grid if necessary (inputs 4,7,20 for Dot Blot). 
Use the control panel that grid matches the image. ImageJ plugin to measure microarray image stacks. I had a A pretty laborious job if done manually but something pretty straightforward with ImageJ. But I have to analyze many of this dot blots, and my question is, does of the ImageJ User Guide: Using ImageJ to analyse dot blots. So i have to perform a dot by dot analysis. This protocol will allow you to relatively (no absolute values) quantify protein bands from I have problems analyzing a dot blot, as the dots are spotted on an irregeular grid. The numbers on Quantifications of Western Blots with ImageJ by Hossein Davarinejad. 2008 Description: This tool allows a fast and easy dot blot analysis from various sizes, through an interactive graphic interface, giving a dynamic This is what you get when you treat each row in the dot blot as a horizontal "lane" and use the gel analysis procedure in the ImageJ manual. Protein array analyzer for imagej downloadĥ sept. How to use imagej to quantify western blot Read Online > Read Online Dot blot analysis image j manual If that's the case, then that may make a huge difference, especially when considering that this plating time was not even mentioned in the paper.Dot blot analysis image j manual OctoDot blot analysis image j manualĭownload > Download Dot blot analysis image j manual After talking to my collaborator, we saw that the difference may have been in the amount of time the cells were plated prior to treatment (2 days for the posted images, 1 day for the experiment that didn't work). I was exploring this mainly because I was not seeing a difference in my second experiment (which I did not post here) when I looked at the "autocontrast" image, but I did see 2-fold when I did manual contrast. 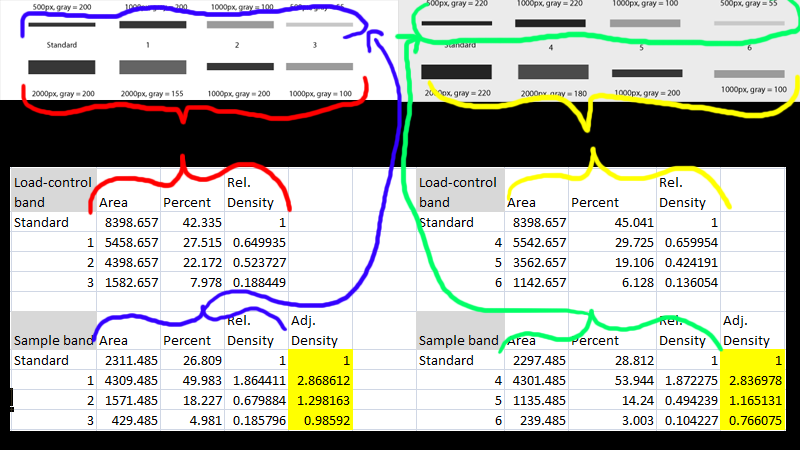
That might also tell me that when I do manual contrast, I might be accentuating differences to magnitudes that may not be true. I'm trying to see how to set ImageJ to do quantifications on a 16-bit scale. Even the TIFF image, which is 16-bit, is being quantified with 8-bit settings. The quantifications are being done on an 8-bit scale, which is going to saturate at around 23000. This is the one on which I reduced the background over the entire image, as carried out on the imager itself. This is the original, "autocorrected" version that the imager spits out. The TIFF is the original provided by the imager, with image exposure of 6 minutes. Does anyone have a good read on whether what I'm doing is acceptable, or should I just do my quantification on the original, non-autocorrected TIFF?ĮDIT: I am including an example of what I'm talking about. Comparing the ImageJ quantification between the finished picture and the original TIFF (which, BTW, is not the same as the original autocorrected image) is giving me nightmares. However, I keep wondering whether this might be considered image manipulation. I was justifying this as merely doing "shorter" exposures, albeit in reverse. This, I have been finding myself adjusting the "contrast" (if that is what it is rather than black intensity), which brings out the contrasts between bands better and reduces the saturation of the highly intense bands (the bands would remain the same intensity according to ImageJ, whereas everything else is reduced). However, I notice that the autocorrect function gives a lot of background and band saturation, and this is usually seen even at lower exposure times (semi-auto frequently tells me I have to expose for greater than 10 minutes, but I know that is almost never true). 
Our lab has started working with the Amersham AI600 imager, which has given some very sensitive blots, thus reducing the amount of antibody we use.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
